|
SinoMab Was Invited to Attend the Inaugural Asia Summit on Global Health
HONG KONG, Nov 26, 2021 - (ACN Newswire) - SinoMab BioScience Limited ("SinoMab", or the "Company", stock code: 3681.HK), a Hong Kong-based biopharmaceutical company dedicated to the research, development, manufacturing and commercialization of therapeutics for the treatment of immunological diseases, was invited to attend the inaugural Asia Summit On Global Health ("ASGH"), jointly organized by the Government of the Hong Kong Special Administrative Region and the Hong Kong Trade Development Council (HKTDC) yesterday, Dr. Shui On LEUNG, Executive Director, Chairman and Chief Executive Officer of SinoMab, joined the panel discussion on the topic of "What's Next for Healthcare Innovation and Investment".
 | | The panel discussion of "What's Next for Healthcare Innovation and Investment";
Dr. Shui On LEUNG, Executive Director, Chairman and Chief Executive Officer of the Company (second right) |
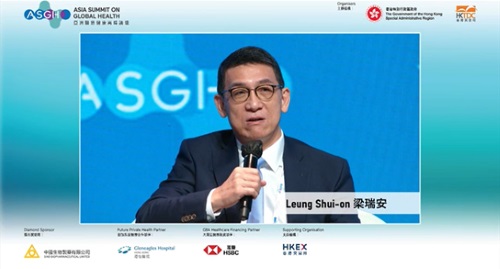 | | Dr. Shui On LEUNG was the guest speaker |
With the theme of "Shaping a Resilient and Sustainable Future", the Inaugural ASGH was held at the Convention Hall of Hong Kong Convention and Exhibition Centre and synchronized online. At the panel discussion with the topic of "What's Next for Healthcare Innovation and Investment", Dr. Shui On LEUNG, Executive Director, Chairman and Chief Executive Officer of the Company, was one of the guest speakers.
Regarding the development of biotechnology and healthcare ecosystem in Hong Kong in recent years, Dr. Shui On LEUNG said that in stead of being a standalone ecosystem, Hong Kong has to merge with either mainland China or other part of the world, and Hong Kong can play the role as a phase I clinical trial center and a trial site. He said that the ecosystem depends on scientific innovation and government involvement. Doing innovative research in Hong Kong enjoys advantages such as legal protection and patenting system, which will facilitate the companies to get licensed with other collaborators. He pointed out that Chapter 18A of the Listing Rules and Guidelines reflects the value of the biotechnology industry, improves market liquidity, and encourages more investment into the biotechnology system.
More than 70 senior government officials, investors, influential business leaders, financial and professional service providers as well as renowned academia from related sectors worldwide were invited to attend the summit to exchange insights and explore business opportunities and partnership. Exhibitions, project pitching and business networking sessions were also be staged to facilitate business negotiation and investment partnerships.
Dr. Shui On LEUNG said, "It is a great honour to attend the Inaugural ASGH on behalf of the Company. Through the communication and sharing with stakeholders including healthcare and technology experts, research and business leader, start-ups, investors and government decision-maker on the summit, the overall development of the ecosystem for the healthcare and biotechnology sectors will be benefited. SM03, the flagship product of the Company, completed the enrolment for Phase 3 clinical trial in this year and expected to readout data next year, indicating that SM03 will soon be commercialized. For business development, we have entered into our first license agreement in September this year. To enhance our R&D and product development capabilities, new plant of over 32,000 liters production capacity of our Suzhou campus is under construction. In the future, the Company will be dedicated to discovering and developing novel drug targets and exploring treatments for immunological diseases, further expanding our product pipeline to fight for patients' well-being. We will also continue to actively looking for opportunities of collaboration and partnership, consolidate the capital market position of the Company as well as contribute to human healthcare and the biotechnology industry of Hong Kong."
About SinoMab BioScience Limited
SinoMab BioScience Limited (stock code: 3681.HK) is dedicated to the research, development, manufacturing and commercialization of therapeutics for the treatment of immunological diseases. The Company's flagship product SM03 is a potential global first-in-target mAb against CD22 for the treatment of rheumatoid arthritis and is currently in Phase III clinical trial for rheumatoid arthritis in China, which has been recognized as one of the significant special projects of Significant New Drugs Development of the Twelfth Five-Year Plan Period and the Thirteenth Five-Year Plan Period. In addition, the Company possesses other potential first-in-target and first-in-class drug candidates, some of which are already in clinical stage, with their indications covering rheumatoid arthritis, systemic lupus erythematosus, pemphigus vulgar, non-Hodgkin's lymphoma, asthma, and other diseases with major unmet clinical needs.
Source: SinoMab BioScience Limited
Sectors: BioTech, Healthcare & Pharm
Copyright ©2026 ACN Newswire. All rights reserved. A division of Asia Corporate News Network. |
Latest Release

SPARX Group Establishes "Mirai Creation Fund IV" Toyota Motor Corporation, Sumitomo Mitsui Banking Corporation, MUFG Bank, Ltd. and Mizuho Bank, Ltd. to Provide Capital Targeting Total Commitments of JPY100 billion
May 12, 2026 19:29 JST
| 
JCB and Credit Card Association of the Philippines (CCAP) Launch Partnership to Boost Financial Literacy Among Filipinos
May 12, 2026 14:00 JST
| 
Aleen Inc. Announces Its OTC Market Listing
May 12, 2026 10:30 JST
| 
Fujitsu Digitalizes Management of Japan's Reserve Self-Defense Force for Ministry of Defense, Enhancing Efficiency
May 07, 2026 14:02 JST
| 
ULVAC Establishes Japan-Based Production for Rare-Earth Magnet Vacuum Melting Furnaces
May 01, 2026 11:00 JST
| 
teamLab Borderless Ranked Among the World's 100 Most Visited Art Museums and 4th in Japan
Apr 30, 2026 21:00 JST
| 
TANAKA to Showcase Advanced Semiconductor Materials and Circular Economy Initiatives at SEMICON Southeast Asia 2026
Apr 28, 2026 21:00 JST
| 
NEC Launches "Africa Corporate Innovation Program" Accelerating Business Through Co-Creation with African Startups
Apr 28, 2026 19:05 JST
| 
Advancing Early Detection: OMRON Healthcare Supports May Measurement Month 2026
Apr 28, 2026 01:00 JST
| 
NEC Announces Strategic Collaboration with Anthropic Focused on Enterprise AI
Apr 23, 2026 17:46 JST
| 
Fujitsu and Carnegie Mellon University launch joint center for Physical AI
Apr 23, 2026 17:19 JST
| 
China Gas Signs Strategic Cooperation Agreement with CITIC Construction and China CITIC Bank
Apr 22, 2026 21:32 JST
| 
CBL International Limited Acquires Majority Stake in Green Marine Energy Holdings Limited
Apr 22, 2026 21:15 JST
| 
OMRON Healthcare and Tricog Health strengthen collaboration in India with Tricog CardioCheck (TCC)
Apr 22, 2026 19:30 JST
| 
MHI Marine Machinery & Equipment and Mitsubishi Shipbuilding Obtain Approval in Principle (AiP) for the Basic Design of a Methane Oxidation Catalyst System for Marine LNG-Fueled Engines from Classification Society ClassNK
Apr 22, 2026 17:16 JST
| 
NEC Receives Company of the Year recognition in Global Biometric Solutions from Frost & Sullivan
Apr 22, 2026 14:46 JST
| 
MHIEC Completes Improvement of Core Equipment at Municipal Solid Waste Incineration Facility in Kagoshima City
Apr 22, 2026 11:13 JST
| 
MHIEC Completes Improvement of Core Equipment at Municipal Solid Waste Incineration Facility in Sendai City
Apr 22, 2026 11:03 JST
| 
Merck & Co., Inc., Rahway, NJ, USA and Eisai Provide Update on Phase 3 LITESPARK-012 Trial Evaluating First-Line Combination Treatments for Certain Patients With Advanced Renal Cell Carcinoma (RCC)
Apr 21, 2026 20:52 JST
| 
Hitachi to establish a new company with Nojima under a strategic partnership to accelerate growth of its home appliance business
Apr 21, 2026 19:21 JST
|
More Latest Release >>
|